 Diamond is among the hardest materials known, while graphite is soft enough to form a streak on paper. For example, diamond is highly transparent, while graphite is opaque and black. The physical properties of carbon vary widely with the allotropic form. This abundance, together with the unique diversity of organic compounds and their unusual polymer-forming ability at the temperatures commonly encountered on Earth, make this element the chemical basis of all known life. It is ubiquitous in all known lifeforms, and in the human body it is the second most abundant element by mass (about 18.5%) after oxygen. It is the 4th most abundant element in the universe by mass after hydrogen, helium, and oxygen. The name "carbon" comes from Latin language carbo, coal, and in some Romance languages, the word carbon can refer both to the element and to coal. Carbon is one of the few elements known to man since antiquity. There are three naturally-occurring isotopes: 12C and 13C are stable, and 14C is radioactive, decaying by 0.158 MeV β - emission with a half-life of about 5700 years.  
It is a group 14, nonmetallic, tetravalent element, that presents several allotropic forms of which the best known ones are graphite (the thermodynamically stable form under normal conditions), diamond, and amorphous carbon. 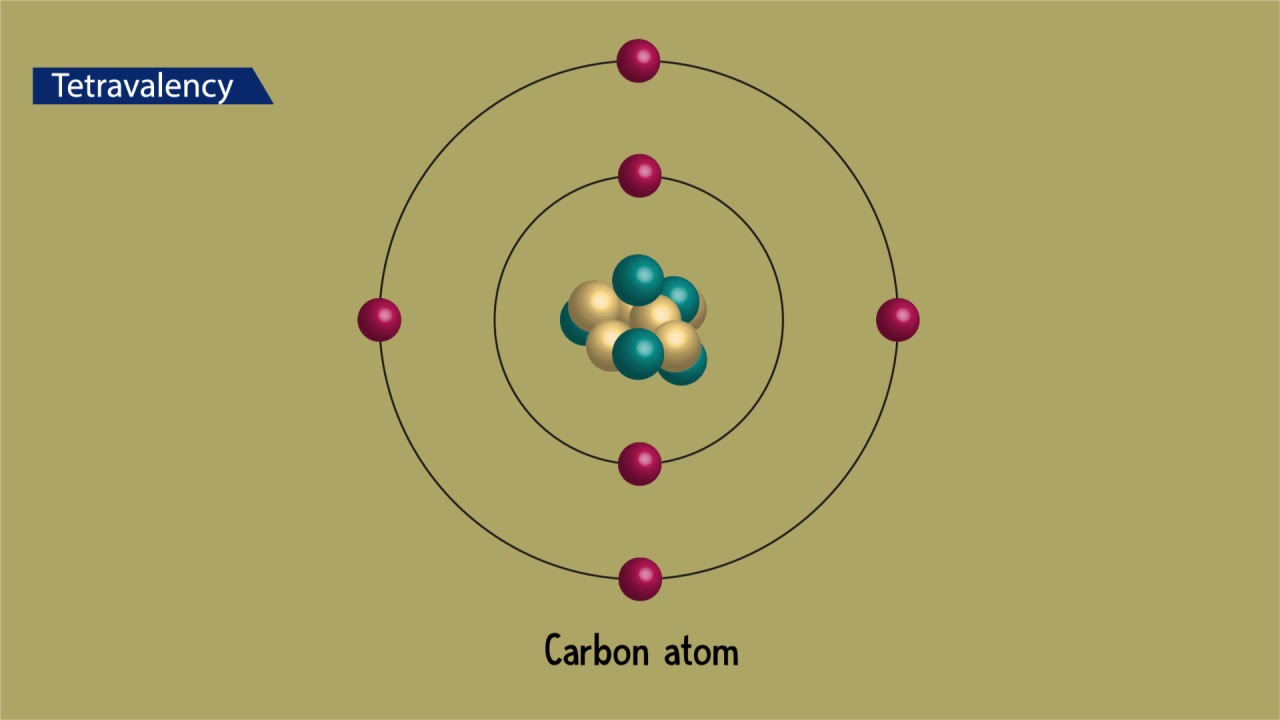
Black (graphite) colorless (diamond) Standard atomic weightĤ, 3, 2, 1, 0, -1, -2, -3, -4 (mildly acidic oxide)Ĭarbon ( pronounced /ˈkɑrbən/) is a chemical element with the symbol C and atomic number 6. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
